
Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other. Take it into your hands, shut your eyes, and twist the sides around randomly a few times. Entropy is a state function that is often erroneously referred to as the 'state of disorder' of a system. This online converter, converts the specific unit of entropy to. The average temperature for the cold water is 21 ° C ( 294 ° K ). Entropy exists to ensure energy conservation and prevent the abuse and misuse of energy. Imagine a 2x2 Rubik's cube, solved so that each face contains just one colour. We estimate entropy change as S Q / T av, where T av is 23 ° C ( 296 ° K ) for the hot water. Saturated Unsaturated and Supersaturated. 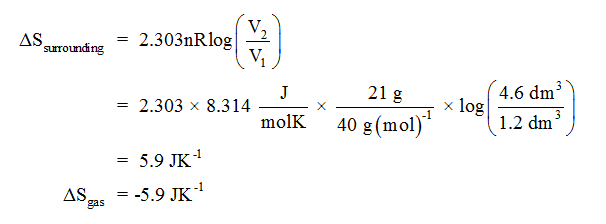
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
